S P Singh Solutions for Chapter: Atomic Structure and Chemical Bonding, Exercise 1: EXERCISE 4(A)
S P Singh Chemistry Solutions for Exercise - S P Singh Solutions for Chapter: Atomic Structure and Chemical Bonding, Exercise 1: EXERCISE 4(A)
Attempt the practice questions on Chapter 4: Atomic Structure and Chemical Bonding, Exercise 1: EXERCISE 4(A) with hints and solutions to strengthen your understanding. Concise Chemistry Class 9 solutions are prepared by Experienced Embibe Experts.
Questions from S P Singh Solutions for Chapter: Atomic Structure and Chemical Bonding, Exercise 1: EXERCISE 4(A) with Hints & Solutions
Name subatomic particle whose charge is:
Which metal did Rutherford select for his -particle scattering experiment and why?
What do you think would be the observation of Scattering experiment if carried out on: (i) Heavy nucleus like platinum (ii) Light nuclei like lithium?
Which part of an atom was discovered by Rutherford?
In the figure given alongside:
Name the shells denoted by , Which shell has the least energy?
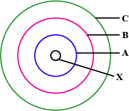
In the figure given alongside:
Name and state charge on it.
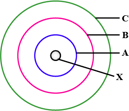
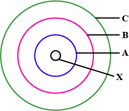
The above sketch is of the _____ model of an atom. (Bohr's / Rutherford's)
Give the postulates of Bohr's atomic model.






