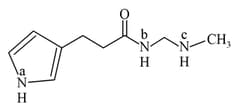
Embibe Experts Solutions for Chapter: Organic Chemistry - Some Basic Principles and Techniques, Exercise 3: Level 3
Embibe Experts Chemistry Solutions for Exercise - Embibe Experts Solutions for Chapter: Organic Chemistry - Some Basic Principles and Techniques, Exercise 3: Level 3
Attempt the practice questions on Chapter 21: Organic Chemistry - Some Basic Principles and Techniques, Exercise 3: Level 3 with hints and solutions to strengthen your understanding. Chemistry Crash Course NEET solutions are prepared by Experienced Embibe Experts.
Questions from Embibe Experts Solutions for Chapter: Organic Chemistry - Some Basic Principles and Techniques, Exercise 3: Level 3 with Hints & Solutions
Among the following.
I 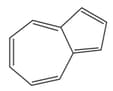
II 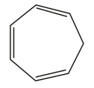
III 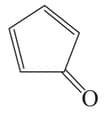
IV 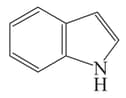
V 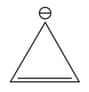
The antiaromatic compounds are
It is difficult to esterify because of:
Two isomeric compounds and are heated with .
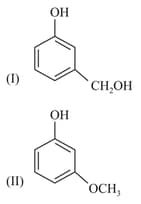
The products obtained are
The ammonia evolved from of a compound in Kjeldahl's estimation of nitrogen neutralizes of solution. The weight percentage of nitrogen in the compound is:
The correct order of the increasing basic nature of the following base is
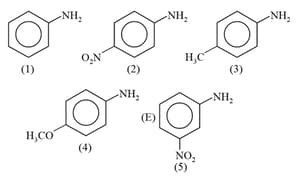
In which of the following pairs of carbocations, the first carbocation is more stable than the second?
() and
() and
() and
() and
Among formic acid, acetic acid, propanoic acid and phenol, the strongest acid in water is:
The most acidic proton and the strongest nucleophilic nitrogen in the following compound respectively, are: