Embibe Experts Solutions for Chapter: Dual Nature of Matter and Radiation, Exercise 1: KCET (UG) 2018
Embibe Experts Physics Solutions for Exercise - Embibe Experts Solutions for Chapter: Dual Nature of Matter and Radiation, Exercise 1: KCET (UG) 2018
Attempt the practice questions on Chapter 30: Dual Nature of Matter and Radiation, Exercise 1: KCET (UG) 2018 with hints and solutions to strengthen your understanding. EMBIBE CHAPTER WISE PREVIOUS YEAR PAPERS FOR PHYSICS solutions are prepared by Experienced Embibe Experts.
Questions from Embibe Experts Solutions for Chapter: Dual Nature of Matter and Radiation, Exercise 1: KCET (UG) 2018 with Hints & Solutions
A proton moving with a momentum has a kinetic energy of its rest mass energy. Another light photon having energy equal to the kinetic energy of the proton possesses a momentum Then the ratio is equal to
The number of photons falling per second on a completely darkened plate to produce a force of is . If the wavelength of the light falling is then
The work function of a metal is . Light of wavelength is incident on this metal surface. The velocity of emitted photoelectrons will be
According to Einstein's photoelectric equation, the graph between kinetic energy of photoelectrons ejected and the frequency of incident radiation is
The maximum kinetic energy of emitted photoelectrons depends on
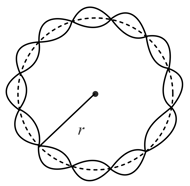
The figure shows standing de Broglie waves due to the revolution of electron in a certain orbit of hydrogen atom. Then the expression for the orbit radius is (all notations have their usual meanings)
An electron in an excited state of ion has angular momentum . The de Broglie wavelength of electron in this state is (where radius). The value of is
A proton and an particle are accelerated through the same potential difference . The ratio of their respective de-Broglie's wavelengths is