PROBLEMS
Ramendra C Mukerjee Chemistry Solutions for Exercise - PROBLEMS
Simple step-by-step solutions to PROBLEMS questions of Solid and Liquid States from Modern Approach to Chemical Calculations. Also get 3D topic explainers, cheat sheets, and unlimited doubts solving on EMBIBE.
Questions from PROBLEMS with Hints & Solutions
Copper has a face-centred cubic structure with a unit-cell edge length of . What is the size of the largest atom (in pm), which could fit into the interstices of the copper lattice without distorting it?
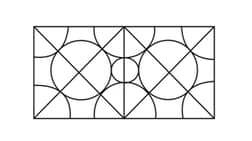
[Hint: Calculate the radius of the smallest circle in the figure.]
A face-centred cubic solid of an element (atomic mass ) has a cube edge of Calculate its density.
A metallic element has a simple cubic lattice. Each edge of the unit cell is The density of the metal is How many unit cells will be present in of the metal?
(Give your answer in multiples of .)
Two capillary tubes of radius and were placed into a sample of liquid The difference between the heights of the liquid in the tubes is . Given that density If , then find .
drops each of water and ether, weigh and , respectively. Determine the surface tension of ether, if the surface tension of water is .
Give an answer to the nearest integer value.
The number of drops of water, counted in the falling-drop method using a stalagmometer is whereas the number of drops of an organic liquid is . The surface tension of an organic liquid is , if the surface tension of water is and the densities of water and the organic liquid are and , respectively. Find the value of .
Round-off and give your answer to the nearest integer value.
The viscosity of olive oil at is and density is . How long will it take to pass through a viscometer, if water under the same conditions takes seconds ?
An organic liquid rises in a capillary tube of radius How much will it rise if, the cross-sectional area of the tube is halved?
