V. K. Sally and R. Goel Solutions for Chapter: Acids, Bases And Salts, Exercise 1: EXERCISE 2A
V. K. Sally Chemistry Solutions for Exercise - V. K. Sally and R. Goel Solutions for Chapter: Acids, Bases And Salts, Exercise 1: EXERCISE 2A
Attempt the practice questions on Chapter 2: Acids, Bases And Salts, Exercise 1: EXERCISE 2A with hints and solutions to strengthen your understanding. Core Science Chemistry solutions are prepared by Experienced Embibe Experts.
Questions from V. K. Sally and R. Goel Solutions for Chapter: Acids, Bases And Salts, Exercise 1: EXERCISE 2A with Hints & Solutions
If a few drops of a concentrated acid accidentally spills over the hand of a student, what should be done?
Sodium hydrogen carbonate when added to acetic acid, evolves a gas. Which of the following statements are true about the gas evolved?
(i) It turns lime water milky.
(ii) It extinguishes a burning splinter.
(iii) It dissolves in a solution of sodium hydroxide.
(iv) It has a pungent order.
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
(i) The temperature of the solution increases
(ii) The temperature of the solution decreases
(iii) The temperature of the solution remains the same
(iv) Salt formation takes place.
An aqueous solution turns red litmus solution blue. Excess addition of which of the following solutions would reverse the change?
Which of the following can be used as an acid-base indicator by visually impaired student?
Pick out the basic substance from the following:
In the following schematic diagram for the preparation of hydrogen gas as shown in the figure what would happen if the following changes are made?
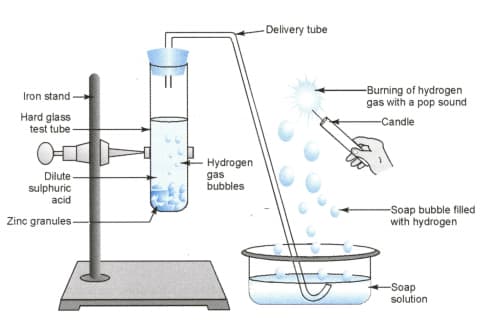
(a) In place of zinc granules, same amount of zinc dust is taken in the test tube
(b) Instead of dilute sulphuric acid, dilute hydrochloric acid is taken
(c) In place of zinc, copper turnings are taken
(d) Sodium hydroxide is taken in place of dilute sulphuric acid and the tube is heated.
And, Which of the following is acidic in nature?
(a)lime juice (b)human blood (c)lime water (d)antacid
(i)Identify the compound X on the basis of the reactions given below. Also write the name and chemical formulae of A, B and C.
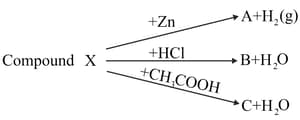
(ii)Which of the following substance will not give carbon dioxide on treatment with dilute acid?
(a)marble, (b) limestone, (c) baking soda, (d) lime.