HARD
Earn 100
At STP, a container has 1 mole of Ar, 2 moles of , 3 moles of and 4 moles of . Without changing the total pressure if one mole of is removed, the partial pressure of is
(a)Changed by about 16%
(b)Halved
(c)Changed by 26%
(d)Unchanged
50% studentsanswered this correctly
Important Questions on States of Matter
MEDIUM
MEDIUM
HARD
(Given: The vapour pressure of water at is )
MEDIUM
The volume of gas is twice than that of gas . The compressibility factor of gas is thrice than that of gas at same temperature. What are the pressures of the gases for equal number of moles?
MEDIUM
MEDIUM
(Given : molar mass and )
MEDIUM
EASY
EASY
EASY
MEDIUM
EASY
[Use atomic masses (in ): ]
MEDIUM
HARD
'' of molecular oxygen is mixed with of neon . The total pressure of the nonreactive mixture of and in the cylinder is bar. The partial pressure of is bar at the same temperature and volume. The value of '' is
[Given: Molar mass of . Molar mass of ]
EASY
EASY
HARD
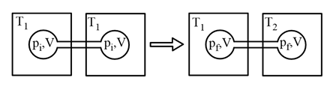
EASY
The above reaction is carried out in a vessel starting with partial pressure , and . When the reaction is complete, the total pressure in the reaction vessel is _____ .
(Round off of the nearest integer).
HARD
EASY

