Describe how to prepare oxygen gas in the lab.

Important Questions on In the Lab
A sample of soil from a vegetable garden was thoroughly crushed, and water added as shown:

Now, using a conical flask, filter funnel, filter paper, universal indicator, and dropping pipette, show how you would measure the of the soil.
A sample of soil from a vegetable garden was thoroughly crushed, and water added as shown:
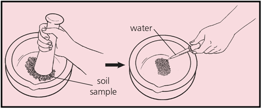
After you measure the of this sample, how would you check that the result for this sample were valid for the whole garden.
Make a drawing of the apparatus labelling the water trough, measuring cylinder, delivery tube, flask, and dropping funnel. This apparatus is used to collect gases in the lab.
This apparatus is used to collect gases in the lab like hydrogen and carbon dioxide, but not sulfur dioxide. Explain why.
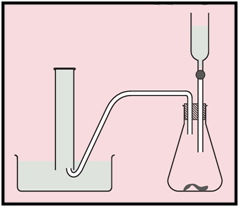
This apparatus is used to measure the rate of a reaction.

Suggest a suitable reagent to use as Y.
This apparatus is used to measure the rate of a reaction.

Which other piece of apparatus is needed?
This apparatus is used to measure the rate of a reaction.

Outline the procedure for this experiment.
This apparatus is used to measure the rate of a reaction.

You must be careful not to use too much of the reagent. Why?
