Draw the Carnot cycle for a monatomic gas on the log-log scale using the - and -coordinates.

Important Questions on Molecular Physics and Thermodynamics
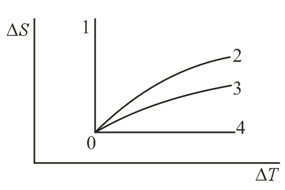
A gas is transferred from an initial state to other states and via different isoprocesses. Which curve representing the dependence of entropy on temperature corresponds to which process?
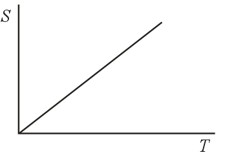
Suppose that the entropy grows linearly with temperature in a process. How does the heat capacity vary with temperature?
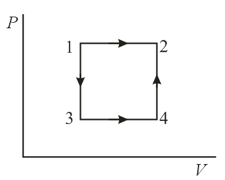
A gas is transferred from a state to a state in two ways: () directly by an isobar, and () first by the isochor ; then by the isobar , and, finally, by the isochor . Show, by direct calculation, that the entropy increment in both cases is the same.
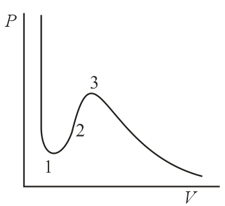
According to the van der Waals equation, which is a third-degree equation in the volume, the theoretical isotherm of a real gas may have either one or three intersections with a horizontal line, the intersections corresponding to either one or three real roots of the equation. With three roots it may so happen that two are equal (maxima and minima on the isotherm) or even all three are equal (the critical point). However, on an isotherm built for a sufficiently low temperature there is a section lying below the horizontal axis, and a horizontal line in this case intersects the section only at two points (two roots in ). Where in this case is the compulsory third root?
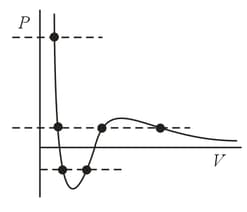
The section on the theoretical isotherm of a real gas (the van der Waals isotherm) is assumed to be unrealistic because of its absolute instability. What is the reason for this instability?