HARD
Earn 100
Draw the structure of cationic detergents
Important Questions on Chemistry in Everyday Life
HARD
EASY
MEDIUM
MEDIUM
EASY
MEDIUM
EASY
EASY
EASY
EASY
EASY
MEDIUM
Match List - I with List - II.
| List-I | List-II | ||
| A | 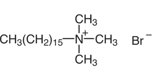 |
I | Dishwashing powder |
| B | 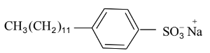 |
II | Toothpaste |
| C | Rosinate | III | Laundry soap |
| D | IV | Hair conditioner |
Choose the correct answer from the options given below
EASY
a)
b) (liq)
c)
d)
EASY
EASY
MEDIUM
EASY
EASY
EASY

