EASY
Earn 100
The correct reaction profile diagram for a positive catalyst reaction.
(a)
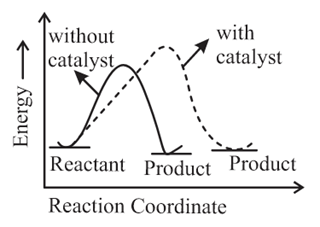
(b)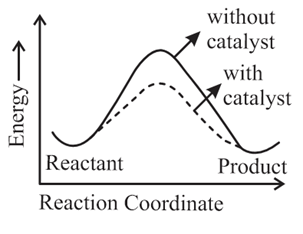

(c)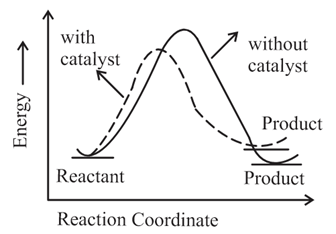

(d)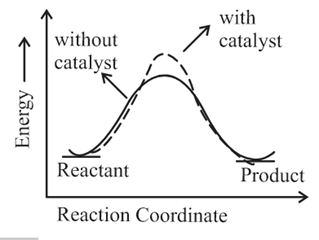

60% studentsanswered this correctly
Important Questions on Chemical Kinetics
HARD
The activation energy of the backward reaction exceeds that of the forward reaction by (in ). If the pre-exponential factor of the forward reaction is times that of the reverse reaction, the absolute value of for the reaction at is ____.
(Given; and is the Gibbs energy)
EASY
MEDIUM
For a reaction, consider the plot of versus given in the figure. If the rate constant of this reaction at is , then the rate constant at is:
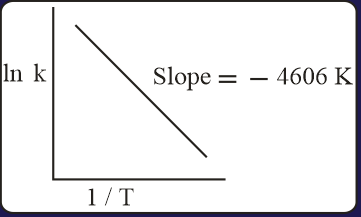
MEDIUM
MEDIUM
EASY
EASY
MEDIUM
EASY
EASY
EASY
MEDIUM
(Assume Activation energy and pre-exponential factor are independent of temperature; )
EASY
MEDIUM
The Arrhenius plots of two reactions, I and II are shown graphically-
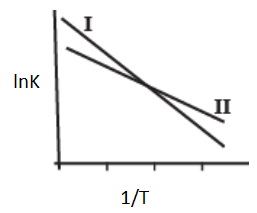
The graph suggests that-
MEDIUM
MEDIUM
EASY
The substances that enhance the activity of a catalyst, are called
MEDIUM
EASY

