The kinetic energy of an electron in the second Bohr orbit of a hydrogen atom is [ is Bohr radius]
Important Questions on Structure of Atom
The difference between the radii of and orbits of is . The difference between the radii of and orbits of is Ratio of is :
| List - I | List - II |
|---|---|
| (I) Radius of the orbit | |
| (II) Angular momentum of the electron in the orbit | |
| (III) Kinetic energy of the electron in the orbit | |
| (IV) Potential energy of the electron in the orbit | |
Which of the following options has the correct combination considering List-I and List-II?
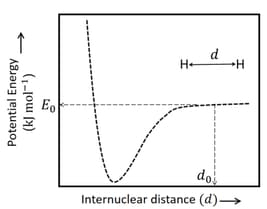
The figure below is the plot of potential energy versus internuclear distance of molecule in the electronic ground state. The value of the net potential energy (as indicated in the figure) is for at which the electron-electron repulsion and the nucleus-nucleus repulsion energies are absent, find the value of to the nearest integer value.
As reference, the potential energy of atom is taken as zero when its electron and the nucleus are infinitely far apart.
Use Avogadro constant as Give an answer to the nearest integer value.
According to Bohr's atomic theory:
(A) Kinetic energy of electron is
(B) The product of velocity (v) of electron and principal quantum number
(C) Frequency of revolution of electron in an orbit is
(D) Coulombic force of attraction on the electron is
Choose the most appropriate answer from the options given below:
(Planck's constant, mass of electron charge of electron permittivity of vacuum, )
| List - I | List - II |
|---|---|
| (I) Radius of the orbit | |
| (II) Angular momentum of the electron in the orbit | |
| (III) Kinetic energy of the electron in the orbit | |
| (IV) Potential energy of the electron in the orbit | |
Which of the following options has the correct combination considering List-I and List-II?

