Which of the following hydrogen bond is strongest in vapour phase?
Important Questions on Chemical Bonding and Molecular Structure
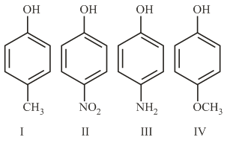
The increasing order of boiling points of the following compounds is :
Consider the following molecules and statements related to them:
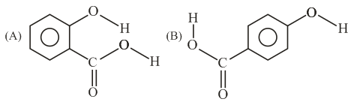
(a) (B) is more likely to be crystaline than (A)
(b) (B) has higher boiling point than (A)
(c) (B) dissolves more readily than (A) in water
Identify the correct option from below:
Given below are two statements:
Statement : -Nitrophenol is steam volatile due to intramolecular hydrogen bonding.
Statement : -Nitrophenol has high melting due to hydrogen bonding.
In the light of the above statements, choose the most appropriate answer from the options given below:
Given below are two statements: one is labelled as Assertion and the other is labelled as Reason .
Assertion : Dipole-dipole interactions are the only non-covalent interactions, resulting in hydrogen bond formation.
Reason : Fluorine is the most electronegative element and hydrogen bonds in are symmetrical.
In the light of the above statements, choose the most appropriate answer from the options given below:





