Oxoacids of Chlorine: Preparation, Structure, Properties and Uses
Oxoacids of Chlorine: Preparation, Structure, Properties and Uses: Overview
This topic covers concepts, such as, Oxoacids of Halogens & Structures of Oxoacids of Chlorine etc.
Important Questions on Oxoacids of Chlorine: Preparation, Structure, Properties and Uses
What products are expected from the disproportionation reaction of hypochlorous acid?
Why does fluorine form only one oxoacid, . Explain.
Identify the chemical formula of oxoacid of halogen, chloric acid.
The correct order of acidity of oxoacids of chlorine is
Identify the name of oxoacid of halogen, .
Write the chemical formula for perchloric acid, hypobromous acid and fluoric acid.
Explain why fluorine does not form oxoacids?
Which oxoacid of chlorine is the most acidic?
Arrange the following :
Increasing order of thermal stability
Increasing acid strength
Increasing reducing nature
Increasing oxidation number of iodine
Increasing acid strength
Increasing oxidising power
Increasing acid strength
Increasing electronegativity
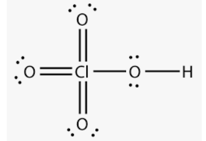
Above structure is of _____ acid.
The structure of is:
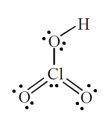
.
Write the different oxoacids of chlorine in the increasing order of their oxidising power.
How would you account for the following?
The oxidising power of oxoacids of chlorine follows the order:
Which is the strongest acid in the following:
Which are the different oxyacids of halogens?
The correct order of acidic strength of following halo acids is-
I.
II.
III.
Among the following, which has maximum acidic character?
The incorrect statement about the oxoacids, and is
Strongest conjugate base is
Strongest conjugate base is