Physical Properties of Aldehydes and Ketones
Physical Properties of Aldehydes and Ketones: Overview
This topic covers concepts such as Physical Properties of Aldehydes and Ketones, Boiling Points Aldehydes and Ketones, Water Solubility of Aldehyde and Ketones, and Odour and Colour of Aldehyde and Ketones.
Important Questions on Physical Properties of Aldehydes and Ketones
The boiling point of aldehyde and ketones are:
Boiling points of aldehydes and ketones are:
The properties of aldehydes and ketones are:
Aldehydes and ketones are differentiated by using:
The solubility of aldehydes and ketones decreases rapidly with increasing length of the alkyl chain. Explain.
Which of the following is a gas at room temperature?
Oil of bitter almonds is
The boiling points of aldehydes and ketones are higher than hydrocarbons and ethers of comparable molecular masses. Explain.
In which of the following solvent(s), aldehydes and ketones are soluble?
Write a note on the solubility of aldehydes and ketones in water as well as in organic solvents.
The boiling points of aldehydes and ketones are higher than hydrocarbons of comparable molecular masses. Give reason.
Lower members of aldehydes and ketones are soluble in water as they can form
An aldehyde molecule, which is a gas at room temperature
Give some information about how the aldehydes and ketones smell and how they impart flavours.
In which of the following options all the given carbonyl compounds are miscible with water in all proportions?
Write a short note on water solubility of aldehyde and ketone.
Decreasing order of boiling point of to follows:
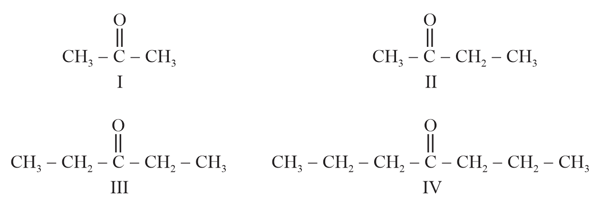
The boiling points of ketones and aldehydes of comparable molecular masses are higher than their corresponding hydrocarbons and ethers because of:
Which among the following has highest boiling point?
Which of the following will have highest boiling point?
