Applications of Grignard Reagent
Applications of Grignard Reagent: Overview
This topic covers concepts, such as Preparation of Alkane by Grignard Reagent, Preparation of Alkane by Grignard Reagent with Active Methylene Group, Preparation of Alkane by Grignard Reagent with 1 Degree Amine, etc.
Important Questions on Applications of Grignard Reagent
on treatment with produces:
𝚆𝚑𝚊𝚝 𝚒𝚜 𝚝𝚑𝚎 𝚊𝚌𝚝𝚒𝚘𝚗 𝚘𝚏 𝚊𝚖𝚖𝚘𝚗𝚒𝚊 𝚘𝚗 𝚐𝚛𝚒𝚐𝚗𝚊𝚛𝚍 𝚛𝚎𝚊𝚐𝚎𝚗𝚝?
Grignard Reagent reacts with primary amine forms alkane.
The preparation of alkane by Grignard Reagent with primary amine is due to the presence of _____.
The reaction of Grignard Reagent with primary amine gives _____.
Write the preparation reaction of alkane by Grignard Reagent with primary amine.
The product obtained when N-methyl ethanamine reacts with ethyl magnesium chloride is _____.
The product obtained when secondary amine reacts with Grignard reagent is _____.
Write the reaction between the secondary amines and Grignard reagent.
Name the product obtained when diethylamine reacts with methyl magnesium chloride.
How does secondary amines react with Grignard reagent?
Which of the following compounds react with ethyl magnesium bromide and also decolourizes bromine water solution?
The product 'A' in the following reaction is 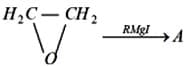
Alcohols react with Grignard reagent to form
In the following reaction B is
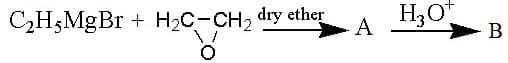
Which one of the following will produce a primary alcohol by reacting with
What is Y in the following sequence of reactions?
A sample of 4.50 mg of unknown alcohol is added to when 1.68 ml of at STP is obtained. The unknown alcohol is
(A); (A) is:
The reaction of 1 mole each of -hydroxyacetophenone and methyl magnesium iodide will give
